PSEB 10th Class Science Chapter 4 Carbon and its Compounds
Table of Contents
MAIN KEY POINTS CH4 CARBON AND ITS COMPOUND
→ In our daily life, the maximum things used by us are compounds of carbon.
→ All living species involve carbon.
→ A very small quantity of carbon present in nature is used by us.
→ Most of the carbon compounds are not good conductors of electricity.
→ The bonding of carbon compounds does not produce any ions.
→ The atomic number of carbon is 6. It has four electrons in its outermost shell.
→ It can gain four electrons forming C4- anion or it can lose four electrons forming C4+ cation.
→ Hydrogen requires one or more than one electron. The shared pair of electrons is said to constitute a single bond between two hydrogen atoms.
→ Chlorine forms a diatomic molecule.
→ Oxygen forms a double bond between two oxygen atoms.
→ In order to attain an octet, each nitrogen atom in a molecule of nitrogen contributes three electrons giving rise to three shared pairs of electrons.
→ Methane is a compound of carbon. It is the major component of bio-gas and compressed natural gas (CNG).
→ It is one of the simplest compounds of carbon.
→ Diamond and graphite are allotropes of carbon. They have the same chemical properties.
→ Fullerenes form another class of carbon allotropes which was first identified as C-60.
→ There are around 3 million carbon compounds in number.
→ The nature of a covalent bond enables carbon to form a very large number of compounds.
→ Carbon shows Catenation to the maximum extent.
→ The organic compounds having the presence of carbon-carbon single bonds only are termed saturated compounds.
→ The carbon-Carbon bond is very stable and strong.
→ Compounds of carbon are formed with oxygen, hydrogen, nitrogen, sulphur, chlorine, and some other elements.
→ Since carbon has a valency of four and is capable of bonding, it forms a large number of compounds.
→ The saturated compounds of carbon and hydrogen are methane, ethane, propane, butane, pentane, hexane.
→ The compounds with identical molecular formulas but different structures are called structural compounds.
→ In addition to straight and branched carbon chains, some compounds have carbon atoms arranged in the form of rings. For example, cyclohexane.
→ Saturated hydrocarbons are called ‘Alkanes’. Such unsaturated hydrocarbons which have one double bond are called ‘Alkenes’. Those having one triple bond are called Alkynes’.
→ All hydrocarbons burn in the presence of oxygen and release heat, light, and CO2.
→ Saturated hydrocarbons generally produce clean flame on burning while unsaturated carbon compounds produce a yellow flame with a lot of black smoke.
→ Incomplete combustion produces a sooty flame.
→ A flame is produced when gaseous substances are burnt.
→ Coal and petroleum are fossil fuels that have been formed from biomass that has been subjected to various biological and geological processes.
→ Alcohol can be converted into carboxylic acids by complete oxidation.
→ Substances that are capable of adding oxygen to other substances are known as oxidizing agents.
→ Catalysts are substances that cause a reaction to occur or accelerate the rate without reacting and getting affected.
→ Ethanol is a liquid at room temperature. Ethanol is commonly called alcohol.
→ Consumption of small quantities of ethanol tends to slow metabolic processes and depress the central nervous system.
→ Consumption of methanol even in very small quantity can cause death. It can also cause blindness by affecting the optic nerve.
→ Methanol is mixed with ethanol to prevent its misuse. Dyes are added to colour the alcohol blue. This is called denatured alcohol.
→ Sugarcane juice can be used to prepare molasses which is fermented to give ethanol.
→ Ethnic acid is commonly called acetic acid. 3.4% solution of acetic acid in water is called vinegar.
→ Carbon: Carbon is an element that is of immense significance in its elemental and combined form. Placed in 14th group of periodic table with atomic number 6, mass number 12 and electronic configuration 4 it has electronic valency 2, 4 and is symbolised as 126C.
→ Carbon compounds: The compounds in which carbon is present are called carbon compounds. These are generally covalent. Ionic compounds of carbonates are not included in this.
→ Covalent bonds: The bond formed by sharing of electrons is known as a covalent bond.
→ Allotropy: The property of an element by virtue of which it can exist in two or more physical forms of the same chemical element, with the same chemical properties.
→ Hydrocarbons: The covalent compounds of hydrogen and carbon are called hydrocarbons.
→ Saturated hydrocarbons: The hydrocarbons with the same chemical formula CnH2n+2 are called alkane. These have a covalent bond between the members of the same category.
→ Isomerism: The compounds with the same molecular formula but different structural formulas are called isomers and the phenomenon is known as isomerism.
→ Unsaturated Hydrocarbons: The compounds of carbon having double or triple bonds between the carbon atoms are known as unsaturated hydrocarbons.
→ Alkene: Generally a hydrocarbon with the chemical formula CnH2n is called alkene.
→ Alkyne: The carbon compound containing a triple bond between carbon atoms is called alkyne.
→ Catenation: The carbon atoms form bonds with other carbon atoms to form long-chained compounds. This property is known as catenation.
→ Allotropy: The phenomenon of the existence of two or more physical forms of the same chemical element, with the same chemical properties, is called allotropy.
→ Fullerenes: There are the allotropes of carbon having a football or bucky ball-like structure.
→ Addition reactions: These are the reactions in which some molecules get added to unsaturated Hydrocarbon like alkene/alkyne.
→ Substitution reactions: These are the reactions in which an atom or group of atoms from an organic compound is replaced by another atom or group.
→ Esterification: The reaction of a carboxylic acid with an alcohol to form an ester is called esterification.
→ Functional group: An atom or group of atoms that determines the chemical properties of an organic compound is called a functional group.
→ Fermentation: The process of conversion of complex organic compounds into simple organic compounds with the help of enzymes is known as fermentation.
→ Alcohol: The compound obtained by the replacement of a hydrogen atom of alkane by hydroxyl (-OH) functional group is called alcohol: Its general formula is CnH2n+1 OH.
→ Carboxylic acid: Carboxylic acids are those organic compounds that have (-COOH) as a functional group.
→ Homologous series: A series of organic compounds having the same functional group formula and possessing similar chemical properties are termed homologous series.
10 PSEB Ch 4 Carbon and its Compounds InText Questions and Answers
Question 1. What would be the electron-dot structure of carbon dioxide which has the formula CO2?
Answer:
In carbon dioxide, carbon atoms are bonded with two oxygen atoms. The atomic number of carbon is 6, and it has four electrons in the outer shell.
To make an octet it requires four electrons. Oxygen requires only two electrons in the outer shell. Therefore electron-dot structure will be :
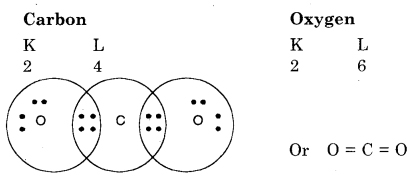
Every oxygen atom is joined to carbon atom by double bond.
Question 2. What would be the electron-dot structure of a molecule of sulphur which is made up of eight atoms of sulphur? (HINT. The eight atoms of sulphur are joined together in the form of ring.)
Answer:
The atomic number of sulphur is 16
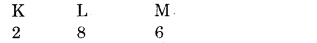
Sulphur has 6 electrons in the outermost shell and to complete an octet it requires 2 electrons.
∴ Sulphur atom will share 2 electrons. It’s chemical formula is S8
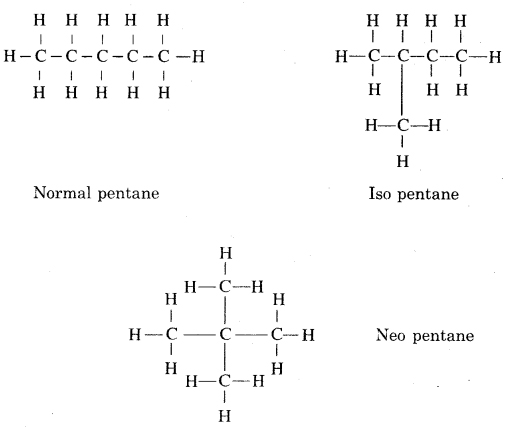
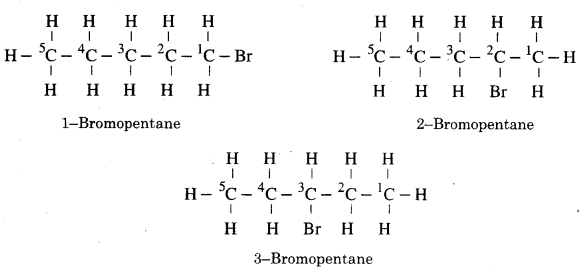
Question 3. How many structural isomers can you draw for pentane?
Answer:
Three ; n-Pentane, iso-pentane, neo-pentane.
Structural isomers can be drawn for pentane.
Question 4. What are the two properties of carbon which lead to the huge number of carbon compounds we see around us?
Answer:
- Catenation: The carbon atoms have an astonishing property to combine and form bond with other carbon atoms to form long chain compounds. This property is known as catenation. In this, either long chain of carbon are in ring form or the carbon atoms join in single, double or triple bond.
- Tetravalency: Carbon has four electrons in the outermost shell. That is why its valency is four and it has got capacity to make bonds with other elements. Oxygen, Hydrogen, Nitrogen, Sulphur, Chlorine and many other elements can make new compounds with the help of carbon.
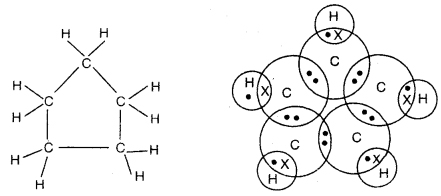
Question 5. What will be the formula and electron dot structure of cyclopentane?
Answer:
Molecular formula of cyclopentane = C5 H2 × 5 = C5H10
Question 6. Draw the structures for following compounds :
Are structural isomers possible for bromopentane?
(i) Ethanoic acid
Answer: Ethanoic acid (CH3COOH)
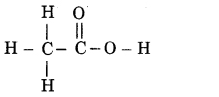
(ii) Bromopentane
Answer:
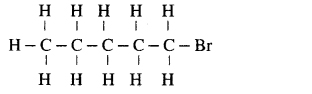
Due to exchange of position of carbon with bromine, many isomers of bromopentane are possible.
For example :
(iii) Butanone
Answer:
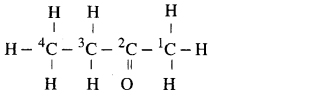
(iv) Hexanal.
Answer
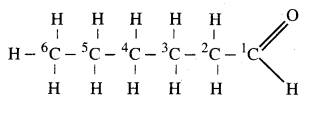
Question 7. How would you name the following compounds?
(i) CH3 – CH2 – Br
Answer:
Bromoethane
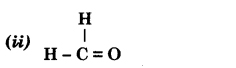
Answer:
Methanal
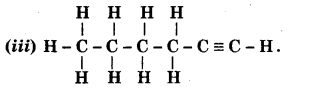
Answer:
Hex-1-yne
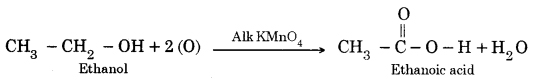
Question 8. Why is the conversion of ethanol to ethanoic acid an oxidation reaction?
Answer:
This is because in this reaction oxygen gets added to ethanol.
Question 9. A mixture of oxygen and ethyne is burnt for welding. Can you tell why a mixture of ethyne and air is not used?
Answer: A mixture of ethyne and air is not burnt for welding. This is because air also contains nitrogen along with oxygen. Nitrogen will also burn in oxygen producing oxides of nitrogen such as nitre oxide (NO) and nitrogen dioxide (NO2) which cause pollution.
Question 10. How would you distinguish experimentally between an alcohol and a carboxylic acid?
Answer: The following two tests are used :
- Litmus test: Treat the given compound with blue litmus solutions. If the blue litmus solution turns red, it is a carboxylic acid and if does not turn red, it is an alcohol.
- Sodium bicarbonate test: Add some sodium bicarbonate solution to the given compound. If their is a brisk evolution of a colourless and odourless gas (CO2) which turns freshly prepared lime water milk, it is carboxylic acid and if their is no effervescence, it is an alcohol.
Question 11. What are oxidising agents?
Answer:
- The substances which can oxidise other substances by giving oxygen are called oxidising agents.
- Examples: Alkaline potassium permanganate solution, acidified potassium dichromate solution, etc.
Question 12. Would you be able to check if water is hard using a detergent?
Answer: No, we can’t check whether the water is hand or soft using a detergent.
Question 13. People use a variety of methods to wash clothes. Usually after adding the soap, they ‘beat’ the clothes on a stone, or beat it with a paddle, scrub with a brush, or the mixture is agitated necessary to get clean clothes?
Answer: This is because when soap molecules dissolve in the dirt, the dirt is somewhat loosened from the clothes, and in order to remove it from clothes, the clothes have to be beaten on a stone or beaten with a paddle or scrubbed with a brush or mixture has to be agitated in washing machines.
Carbon and its Compounds Textbook Questions and Answers
Question 1. Ethane, with the molecular formula C2H6 has :
(a) 6 covalent bonds
(b) 7 covalent bonds
(c) 8 covalent bonds
(d) 9 covalent bonds.
Answer:
(b) 7 covalent bonds
Question 2. Butanone is a four-carbon compound with the functional group :
(a) carboxylic acid
(b) aldehyde
(c) ketone
(d) alcohol.
Answer:
(c) ketone
Question 3. While cooking, if the bottom of the vessel is getting blackened on the outside, it means that :
(а) the food is not cooked completely
(b) the fuel is not burning completely
(c) the fuel is wet
(d) the fuel is burning completely.
Answer:
(b) the fuel is not burning completely
Question 4. Explain the nature of the covalent bond using the bond formation in CH3Cl.
Answer: The formation of CH3Cl can be represented as :
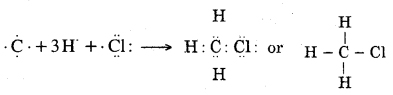
Carbon forms single covalent bonds with three H- atoms and one Cl – atom by sharing one electron pair with each C-H bonds are non-polar. But C – Cl bond is polar because C and H leave almost same electronegativity whereas Cl has more electronegativity than carbon.
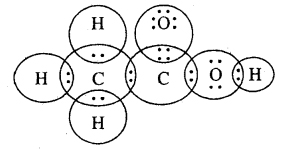
Question 5. Draw the electron dot structures for :
(a) ethanoic acid
Answer:
(b) H2S
Answer:
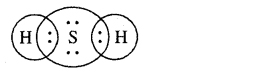
(c) propanone
Answer:
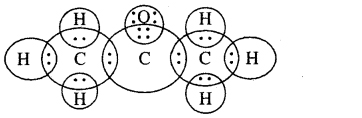
(d) F2.
Answer:

Question 6. What is an homologous series? Explain with an example.
Answer: A series of compounds having similar structural formulae, same functional group and hence similar chemical properties is called a homologous series. In the homologous series any two adjacent members differ by CH2 unit in their molecular formulae.
For example homologous series of aldehydes (or alkanals) can be represented as :
| H – CHO | Methanal |
| CH3 – CHO | Ethanal |
| CH3 – CH2 – CHO | Propanal |
| CH3 – CH2 – CH2 – CHO | Butanal |
| CH3 – CH2 – CH2 – CH2 – CHO | Pentanal and so on. |
Question 7. How can ethanol and ethanoic acid be differentiated on the basis of their physical and chemical properties?
Answer:
Differences between ethanol and ethanoic acid
| Ethanol | Ethanoic acid |
| 1. It is a colourless liquid having a pleasant smell. | 1. It is colourless liquid having vineger like smell. |
| 2. It has no action with a litmus solution. | 2. It turns blue litmus solution red. |
| 3. It has no action with sodium hydrogen carbonate solution. | 3. It decomposes sodium hydrogen carbonate solution giving brisk effervescence of carbon dioxide gas. |
Question 8. Why does micelle formation take place when soap is added to water? Will a micelle be formed in other solvents such as ethanol also?
Answer: Soap molecule has two ends, one is hydrophilic, and it dissolves in water, while the other end is hydrophobic, and it dissolves in hydrocarbons. When soap is at the surface of water , the hydrophobic ‘tail’ of soap will not be soluble in water and the soap will align along the surface of water with the ionic end in water and the hydrocarbon ‘tail’ pointing out of water.
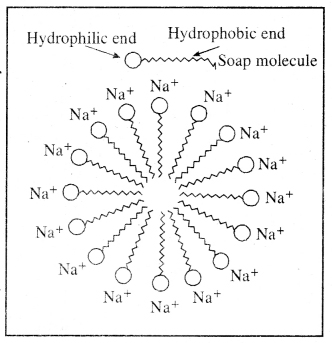
Inside water, these molecules have a unique orientation which keeps the hydrocarbon portion out of the water. This is achieved due to the formation clusters of molecules in which the hydrophobic tails are in the interior of the cluster and the ionic ends are on the surface of the cluster. This formation is called a micelle.
Such micelles can be formed in other polar solvents like ethanol.
Question 9. Why are carbon and its compounds used as fuels for most applications?
Answer: Carbon and its compounds are used as fuels for most applications because they bum producing a large amount of heat and light.
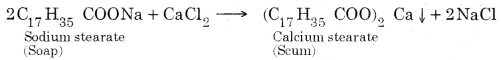
Question 10. Explain the formation of scum when hard water is treated with soap.
Answer: When soap is added to hard water, the soluble calcium and magnesium salts present in it react with soap to give insoluble calcium salt of soap which produces scum.
Question 11. What change will you observe if you test soap with litmus paper (red and blue)?
Answer: Soap solution will turn red litmus paper blue because soap is alkaline in nature.
Question 12. What is hydrogenation? What is its industrial application?
Answer: The addition of hydrogen to unsaturated hydrocarbons in the presence of catalysts like palladium, platinum, nickel etc. to give saturated hydrocarbons is called hydrogenation.
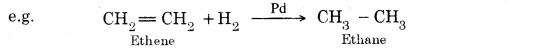
This reaction is used for hydrogenation of liquid vegetable oils using a nickel catalyst to get artificial or vanaspati ghee.
Question 13. Which of the following hydrocarbons undergo addition reaction : C2H6, C3H8, C3H6, C2H2 and CH4.
Answer:
Out of C2H6, C3H8, C3H6, C2H2 and CH4; C3H6 and C2H2 undergo addition reactions because they contain multiple bonds.
Question 14. Give a test that can be used to differentiate chemically between butter and cooking oil.
Answer: Distinction between Butter and Cooking oil:
| Butter | Cooking Oil |
| 1. It is solid at room temperature. | 1. It is liquid at room temperature. |
| 2. Mix equal volumes of HgCl2 solution in 50% alcohol and 5% iodine solution in alcohol. To this add lg of butter. Violet colour does not fade away. | 2. Mix equal volumes of HgCl2 solution in 50% alcohol and 5% solution of iodine in alcohol. To this add 1 ml of cooking oil. Violet colour fades away. |
| 3. Take 2 g of butter in a test tube. To this add 1ml of cone. HCl and a few drops of 2% furfural solution in alcohol. Shake and allow to stand for 5 – 10 minutes. No rose red coloration appears. | 3. Take 2 g of cooking oil in a test tube. To this add 1 ml of cone. HCl and a few drops of 2% .furfural solution in alcohol. Shake it and allow to stand for 5 – 10 minutes. Rose red coloration is obtained. |
Question 15. Explain the mechanism of the cleansing action of soaps.
Answer: Mechanism of cleansing action of Soap:
Soaps are sodium or potassium salts of higher fatty acids e.g. sodium palmitate, C15H31COO–Na+, sodium stearate, C17H35COO–Na+ etc. A molecule of soap consists of two parts :
- a long chain hydrocarbon part (C15H31–, C17H35 …. etc.) which is soluble in oil and
- ionic part on polar group, – COO–Na+ which is soluble in water. Thus a molecule of soap can be represented as :
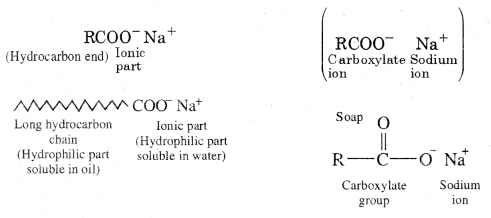
The long hydrocarbon chain is insoluble in water but soluble in oil and greases whereas the ionic or polar part is soluble in water. Soap has a capacity to clean a dirty piece of cloth whereas ordinary water cannot. The dirty clothes contain greasy and oily substance (dirt). Soap molecules dissociate in water to give carboxylate ion (RCOO–) and cation (Na+). When soap added to dirty clothes dipped in water, the hydrocarbon part of carboxylate group dissolving in greasy or oily dirt particles where the polar (COO–) group remain attached to water. In this way each oil droplet acquires negative charge.
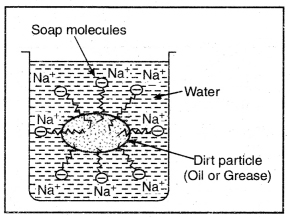
The cleansing action of Soap
These negative charged oil droplets called micelles cannot coalesce and hence form a stable emulsion water. These small droplets along with dirt can be easily washed away with water. Thus the soap helps in removing greasy dirt by producing a stable oil in wrater type emulsion. Also the soap reduces surface tension of water. Hence cloth is wetted more effectively and is cleaned
MCQ Chapter 4 Carbon and its Compounds Multiple Choice Questions:
Question 1. Ethanoic acid has the formula:
(A) C2H5OH
(B) CH3COCH3
(C) CH3COOH
(D) C2H5COOH.
Answer:
(C) CH3COOH
Question 2. Carboxylic acids contain functional group:
(A) -CHO
(B) -CH2OH
(C) -COOH
(D) -OH.
Answer:
(C) -COOH
Question 3. The general formula of the alkyne is:
(A) CnH2n-2
(B) CnH2n+2
(C) CnH2n
(D) CnH2n+2.
Answer:
(A) CnH2n-2
Question 4. Propanone has the functional group :
(A) -OH
(B) -CHO
(C) C=O
(D) -COOH.
Answer:
(C) C=O
Question 5. Vinegar contains acetic acid:
(A) 5-8%
(B) 15-20%
(C) 21-29%
(D) 30-40%.
Answer:
(A) 5-8%
Question 6. The reaction of acetic acid with ethyl alcohol is called :
(A) Polymerisation
(B) Saponification
(C) Hydrogenation
(D) Esterification.
Answer:
(D) Esterification.
Question 7. Propanone has :
(A) 7 covalent bonds
(B) 8 covalent bonds
(C) 9 covalent bonds
(D) 10 covalent bonds.
Answer:
(D) 10 covalent bonds.
Very Short Answer Type Questions
Question 1. Which element is present necessarily in the following items?
Food, clothes, medicines and hooks.
Answer: Carbon.
Question 2. On which element do all living processes depend?
Answer: Carbon.
Question 3. What percentage of carbon is present in minerals found under the crust of earth?
Answer: 0.02 %.
Question 4. What percentage of CO2 is present in atmosphere?
Answer: 0.003%.
Question 5. How many electrons are present in the outer shell of carbon?
Answer: 4.
Question 6. Why does carbon need four electrons to gain or lose?
Answer: To attain noble gas configuration.
Question 7. How does carbon gain four electrons?
Answer: By making C4– anion.
Question 8. How does carbon lose four electrons?
Answer: By making C4+ cation.
Question 9. Which type of bond is shared by a pair of two hydrogen atoms?
Answer: Single bond.
Question 10. How is single bond between two atoms expressed?
Answer: By single straight line.
Question 11. What is the atomic number of nitrogen?
Answer: 7.
Question 12. How many electrons are given by each atom of Nitrogen to complete the octet?
Answer: 3.
Question 13. Which gas is used as a fuel the most?
Answer: Methane gas.
Question 14. Methane is the main constituent gas of which two fuel gases?
Answer: Biogas, C.N.G.
Question 15. Write the full form of C.N.G.
Answer: Compressed Natural Gas.
Question 16. How is the structure of diamond?
Answer: Strong three-dimensional structure.
Question 17. How is the structure of graphite?
Answer: Hexagonal.
Question 18. Which of these two is a good conductor of electricity diamond or graphite?
Answer: Graphite.
Question 19. Which is the hardest substance?
Answer: Diamond.
Question 20. How does it feel by touching graphite?
Answer: Soft and slippery.
Question 21. Which element has the ability to form large number of compounds?
Answer: Carbon.
Question 22. Give names of two elements which are placed in the group of carbon and have same valency.
Answer: Valency of carbon is 4. This group has silicon and germanium.
Question 23. What is the simplest form of hydrocarbon?
Answer: Methane (CH4).
Question 24. In which types of bonds can carbon atoms join?
Answer: Single, double, triple bond and ring-shaped.
Question 25. Write the names of five elements which can make bond with carbon to form new compounds.
Answer: Oxygen, Hydrogen, Nitrogen, Sulphur, Chlorine.
Question 26. Write the formula of Ethane.
Answer: C2H6.
Question 27. Give the name and formula of acid present in vinegar.
Answer: Ethanoic acid (CH3 COOH).
Question 28. Give the next higher homo-logues of:
(i) C3H6
Answer: C4H8
(ii) C6H8.
Answer: C7H10.
Question 29. Give name of four carbon compounds.
Answer: Methane, Chloroform, Ethanol and ethanoic acid.
Question 30. Give two allotropic forms of carbon.
Answer: Diamond and graphite.
Question 31. Give the name of simplest ketone.
Answer: Propanone (CH3 COCH3).
Question 32. Give the name of first member of homologous series of alkynes.
Answer: Ethyne (C2H2).
Question 33. Name the products obtained when ethanol undergoes complete combustion.
Answer: Carbon dioxide (CO2) and Water (H2O).
Question 34. What is the atomic number of hydrogen?
Answer: One.
Question 35. Give the electronic configuration of carbon atom.
Answer: (2, 4).
Question 36. Which of the following compounds belong to same homo-Iogous series?
C2H6O2, C2H6O2, C2H6, CH4O
Answer:
C2H6 O, (C2H5OH) And CH4O or (CH2OH).
Question 37. Give three uses of ethanoic acid.
Answer:
- In the manufacture of vineger
- In the manufacture white lead
- As a reagent in laboratory.
Question 38. What is denatured spirit or alcohol?
Answer: It is prepared by adding poisonous substance like methanol, acetone, pyridine or copper sulphate to ethanol. It is unfit for drinking purposes.
Question 39. Define a Catalyst.
Answer: It is a substance which can increase the rate of a reaction but remains unchanged in mass and composition at the end of reaction.
Question 40. Define covalent bond.
Answer: It is formed by mutual sharing of electron pair between two atoms.
Question 41. Explain what covalent compounds do not conduct electric current?
Answer: This is because there are neither free ions nor free electrons.
Question 42. Name the bond formed between metal atom and non-metal atoms.
Answer: Ionic or electro valent bond.
Question 43. What are allotropes?
Answer: When an element exists in two or more form having different physical pruperties but, some chemical properties, there forms are called allotropes.
Question 44. Define catenation.
Answer: It is the property due to which a large number of atoms of the same element get linked together through covalent bonds forming long straight chains, branched chains and rings of different sizes.
Question 45. Give two oxidising agents.
Answer:
- Acidified potassium permanganate.
- Acidified potassium dichromate.
Question 46. Define functional group.
Answer: It is an atom or group of atoms which when present in a molecule gives special properties to it. e.g. C2H5OH has the functional group, -OH (hydroxyl group).
Question 47. If two organic compounds have some functional group, predict they have similar physical or chemical properties.
Answer: Similar chemical properties.
Question 48. Give the general formulae of alkanes, alkenes and alkynes.
Answer:
Alkanes: Cn H2n+2
Alkaenes: CnH2n
Alkynes: CnH2n-2, where n = No. if carbon atoms).
Question 49. Give the first four members of homologous series of alcohols.
Answer: CH3OH, C2H5OH. C3H7OH, C4H5OH.